Buy Bupivacaine Injection Online
Looking to buy bupivacaine injection? This is your definitive guide for healthcare professionals. Explore comprehensive information on bupivacaine hydrochloride injection uses, different concentrations like bupivacaine 0.25 2.5 mg ml injection and bupivacaine 0.5 injection, and the correct bupivacaine injection cpt code. Learn how to safely order bupivacaine injection online and discover the best place to order bupivacaine injection. We provide clear guidance on how to buy bupivacaine injection in USA and specifically how to order bupivacaine injection in Colorado, ensuring your bupivacaine injection buy is safe, compliant, and effective for optimal patient care.
The Ultimate Guide to Bupivacaine Injection: Uses, Safety, and Procurement
Comprehensive Introduction to Bupivacaine Hydrochloride Injection
In the landscape of modern anesthesiology and pain management, few medications have proven as valuable and versatile as bupivacaine hydrochloride injection. As a long-acting local anesthetic belonging to the amide class, bupivacaine has become an indispensable tool for healthcare providers across various medical specialties. Its ability to provide prolonged sensory blockade with minimal motor interference makes it particularly valuable for surgical procedures, post-operative pain management, and diagnostic interventions. For medical professionals seeking to buy bupivacaine injection, understanding its pharmacological profile, clinical applications, and safety considerations is paramount to ensuring optimal patient outcomes.
The evolution of bupivacaine since its introduction has been marked by continuous research into its mechanisms of action, formulation improvements, and expanded clinical applications. Today, healthcare providers can access various concentrations and formulations, including bupivacaine 0.25 2.5 mg ml injection, bupivacaine 0.5 injection, and combination products such as bupivacaine and kenalog injection or bupivacaine and triamcinolone injection. Each formulation serves specific clinical purposes, tailored to the duration and intensity of anesthesia required for different procedures. This comprehensive guide serves as an authoritative resource for clinicians, pharmacists, and healthcare administrators who need to order bupivacaine injection and implement it safely in their practice settings.
For medical facilities and practitioners looking to order bupivacaine injection online, this guide provides essential information on proper usage, safety protocols, and regulatory considerations. We will explore the critical factors that influence the decision to bupivacaine injection buy, including quality standards, storage requirements, and legal procurement processes. Whether you’re seeking to buy bupivacaine injection in USA or specifically need to order bupivacaine injection in Colorado, this resource offers the specialized knowledge needed to navigate the procurement landscape while maintaining compliance with medical standards and regulations.
Pharmacological Profile and Mechanism of Action
Bupivacaine hydrochloride is chemically designated as (±)-1-Butyl-N-(2,6-dimethylphenyl)–2-piperidinecarboxamide monohydrochloride monohydrate, a white crystalline powder that is freely soluble in 95 percent ethanol and soluble in water. The pharmacological properties of bupivacaine stem from its ability to block the generation and conduction of nerve impulses by increasing the electrical excitation threshold for neuronal membranes, slowing the rise of action potential, and reducing the rate of depolarization. This mechanism effectively prevents the initiation and transmission of nerve impulses, producing the characteristic anesthetic effect.
The duration of action with bupivacaine is significantly longer than that of other local anesthetics such as lidocaine or mepivacaine, making it particularly valuable for procedures requiring extended pain control. The half-life of bupivacaine hydrochloride in adults is approximately 2.7 hours, while in neonates, it extends to 8.1 hours, indicating age-related pharmacokinetic differences that must be considered when determining dosage.
In clinical studies, elderly patients have been observed to reach the maximal spread of analgesia and maximal motor blockade more rapidly than younger patients, with elderly patients also exhibiting higher peak plasma concentrations following administration of this product. The total plasma clearance was decreased in the elderly population, underscoring the need for dose adjustments in this demographic.
Bupivacaine’s protein binding properties are also noteworthy, with approximately 95% of the drug bound to plasma proteins, primarily alpha-1 acid glycoprotein. Factors influencing plasma protein binding, such as acidosis, systemic diseases which alter protein production, or concomitant medications that compete for binding sites, can significantly impact the free fraction of the drug and consequently its efficacy and toxicity profile.
This high degree of protein binding contributes to bupivacaine’s prolonged duration of action but also creates potential for drug interactions that must be carefully considered when planning anesthetic regimens.
Clinical Applications and Indications
Bupivacaine Hydrochloride Injection is indicated in adults for the production of local or regional anesthesia or analgesia for surgery, dental and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures.
For each type of block indicated to produce local or regional anesthesia or analgesia, specific concentrations and presentations are recommended. The versatility of bupivacaine makes it suitable for various anesthesia techniques, including infiltration, peripheral nerve block, sympathetic nerve block, epidural block, caudal block, and retrobulbar block.
In surgical settings, bupivacaine is frequently employed for procedures requiring prolonged anesthesia, such as orthopedic surgeries, abdominal procedures, and thoracic operations. The bupivacaine injection 0.5 concentration is commonly used for peripheral nerve blocks, providing extended postoperative pain relief that reduces the need for systemic analgesics. For less intensive procedures or when a shorter duration of action is desired, clinicians may opt for bupivacaine 0.25 2.5 mg ml injection, which offers effective anesthesia with a slightly reduced duration of effect.
In pain management clinics, bupivacaine is often used in combination with corticosteroids for therapeutic injections. Formulations such as bupivacaine and kenalog injection (containing triamcinolone acetonide) or bupivacaine and triamcinolone injection are commonly administered for joint injections, trigger point injections, and other pain management procedures. These combination products leverage the anesthetic properties of bupivacaine for immediate pain relief and the anti-inflammatory effects of corticosteroids for prolonged benefit, making them valuable tools in managing chronic pain conditions.
For obstetrical anesthesia, bupivacaine has been widely used for epidural analgesia during labor and delivery. However, specific precautions must be observed, as there have been reports of cardiac arrest with difficult resuscitation or death during use of bupivacaine hydrochloride for epidural anesthesia in obstetrical patients. In most cases, this has followed use of the 0.75% (7.5 mg per mL) concentration, which is not recommended for obstetrical anesthesia and should be reserved for surgical procedures where a high degree of muscle relaxation and prolonged effect are necessary.
Dosage Forms and Concentrations
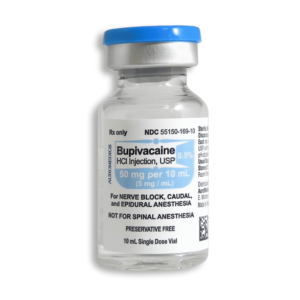
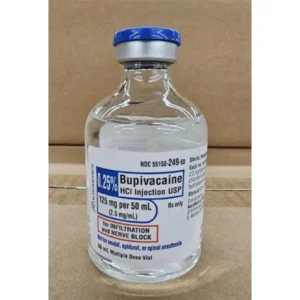
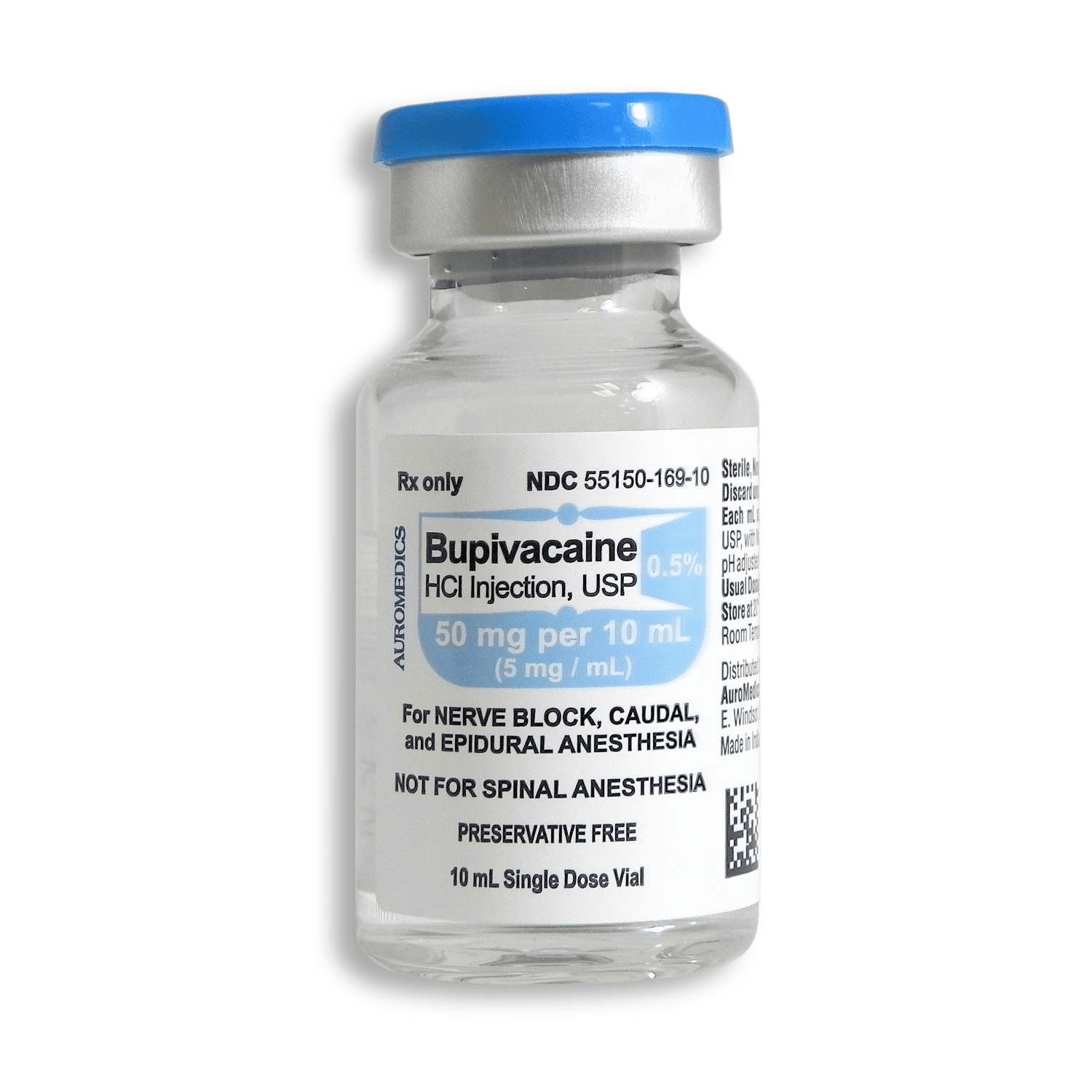
Bupivacaine is available in multiple concentrations and formulations to accommodate various clinical needs. The most commonly used concentrations include 0.25% (2.5 mg/mL), 0.5% (5 mg/mL), and 0.75% (7.5 mg/mL) solutions. The bupivacaine 0.25 2.5 mg ml injection is often used for infiltration anesthesia and peripheral nerve blocks where moderate duration of action is sufficient. The bupivacaine injection 0.5 concentration provides a longer duration of anesthesia and is frequently used for epidural anesthesia, major peripheral nerve blocks, and procedures requiring more profound and extended sensory blockade.
For specific clinical situations, bupivacaine is formulated with other agents to enhance its therapeutic effects. The bupivacaine and epinephrine injection combination contains a vasoconstrictor that delays systemic absorption, prolongs the duration of action, and reduces systemic toxicity. This formulation is particularly useful for techniques where vasoconstriction is advantageous, such as infiltration anesthesia in highly vascular areas. Another specialized formulation is bupivacaine dextrose injection or bupivacaine hydrochloride in dextrose injection, which is designed for spinal anesthesia. The hyperbaric solution containing dextrose allows for better control of the spread of anesthesia within the subarachnoid space.
The selection of appropriate concentration and formulation depends on various factors, including the type of block, desired duration of anesthesia, patient characteristics, and specific procedural requirements. Healthcare providers must carefully consider these factors when determining which formulation to use for each clinical situation. For those looking to order bupivacaine injection online, understanding these different formulations is essential to ensure that the appropriate product is selected for each clinical application.
Safety Considerations and Contraindications
The administration of bupivacaine requires careful attention to safety considerations and contraindications to prevent adverse outcomes. One of the most significant concerns is the risk of systemic toxicity, particularly cardiac toxicity, which can occur with unintentional intravascular injection or excessive dosing. There have been reports of cardiac arrest with difficult resuscitation or death during use of bupivacaine hydrochloride for epidural anesthesia in obstetrical patients, particularly when using the 0.75% concentration.
Unintended intravascular or intrathecal injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progressing ultimately to respiratory arrest.
To minimize this risk, healthcare providers should always perform aspiration tests before injecting bupivacaine and administer the drug slowly and incrementally, especially when using higher concentrations. The injection procedures require the utmost care, and injections should be made slowly and with frequent aspirations to avoid rapid intravascular injection which could lead to high plasma levels and serious adverse effects.
Bupivacaine is contraindicated in patients with known hypersensitivity to bupivacaine or any amide-type local anesthetic. It should also be avoided in patients with severe hypotension, such as cardiogenic or hypovolemic shock. The 0.75% concentration is specifically contraindicated for obstetrical anesthesia and should not be used for peripheral nerve blocks or intravenous regional anesthesia (Bier block) due to the increased risk of severe adverse reactions.
Additional contraindications include paracervical block anesthesia in obstetrics, and any situation where the use of the drug is deemed medically inadvisable. Healthcare providers must carefully evaluate patient history, including any previous adverse reactions to local anesthetics, before deciding to order bupivacaine injection for use in their practice. A thorough understanding of these contraindications is essential for any medical professional looking to bupivacaine injection buy for clinical use.
Dosage and Administration Guidelines
Proper dosage and administration of bupivacaine are critical to achieving effective anesthesia while minimizing the risk of adverse effects. The recommended dose of bupivacaine hydrochloride injection depends on the type of block, the vascularity of the tissues, the number of segments to be blocked, the degree of anesthesia required, individual tolerance, and the physical condition of the patient.
For adult patients, the maximum recommended single dose of bupivacaine hydrochloride without epinephrine is 175 mg, while with epinephrine it increases to 225 mg. The maximum recommended daily dose is 400 mg. For epidural anesthesia in adults, the typical dose ranges from 15-30 mL of 0.25%-0.5% solution, while for caudal anesthesia, 15-30 mL of 0.25%-0.5% solution is recommended. For peripheral nerve blocks, the dosage varies depending on the specific nerve and the extent of anesthesia required.
When administering bupivacaine 0.25 2.5 mg ml injection, healthcare providers should adjust the volume according to the specific needs of the procedure and the patient’s characteristics. For more intensive procedures requiring prolonged anesthesia, the bupivacaine injection 0.5 concentration may be preferred, with careful attention to not exceeding the maximum recommended dose.
For pediatric patients, dosage calculations must be based on weight, with careful consideration of the child’s age and physical condition. The recommended dose for pediatric caudal anesthesia is 0.75-1.25 mL/kg of 0.25% solution, not to exceed 30 mL. For peripheral nerve blocks in children, the dose should be carefully calculated and adjusted based on the specific procedure and the child’s weight.
It is essential to remember that these are general guidelines, and individual patient factors may necessitate dose adjustments. Healthcare providers must use their clinical judgment and experience to determine the appropriate dose for each patient. For those who order bupivacaine injection in Colorado or any other location, understanding these dosage guidelines is crucial for safe clinical practice.
Adverse Reactions and Management
Despite its efficacy, bupivacaine can cause adverse reactions that healthcare providers must be prepared to recognize and manage promptly. Adverse reactions to bupivacaine are generally dose-related and may result from high plasma levels caused by excessive dosage, rapid absorption from the injection site, diminished tolerance, or unintentional intravascular injection.
The most serious adverse reactions involve the central nervous and cardiovascular systems. CNS reactions may include apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred vision, vomiting, sensations of heat, cold, or numbness, twitching, tremors, convulsions, unconsciousness, and respiratory depression. Cardiovascular reactions may include hypotension, hypertension, bradycardia, arrhythmias, and cardiac arrest.
Allergic reactions to bupivacaine are rare but can occur, manifesting as dermatologic reactions, edema, or anaphylaxis. Healthcare facilities that buy bupivacaine injection should have emergency equipment and medications readily available to manage these potential reactions.
Management of bupivacaine toxicity requires immediate recognition and intervention. For CNS toxicity, maintaining a patent airway and administering oxygen are priorities. Benzodiazepines may be used to control seizures. For cardiovascular toxicity, advanced cardiac life support protocols should be initiated immediately, with particular attention to managing arrhythmias and maintaining perfusion. Intralipid emulsion therapy has been recommended for the treatment of local anesthetic systemic toxicity and should be available in facilities where bupivacaine is administered.
Special Populations and Considerations
Certain patient populations require special considerations when receiving bupivacaine. Elderly patients may be more sensitive to the effects of bupivacaine and may require reduced doses due to decreased hepatic function and protein binding. In clinical studies, elderly patients have been observed to reach the maximal spread of analgesia and maximal motor blockade more rapidly than younger patients, with elderly patients also exhibiting higher peak plasma concentrations following administration of this product.
Pediatric patients also require special attention, with dosage calculations based on weight and careful monitoring for signs of toxicity. The pharmacokinetics of bupivacaine in children differ from adults, with a longer half-life observed in neonates (8.1 hours) compared to adults (2.7 hours).
Pregnant patients present another special consideration. Bupivacaine crosses the placenta, and its use during labor and delivery requires careful attention to dosage and timing. The 0.75% concentration is contraindicated for obstetrical anesthesia due to reports of cardiac arrest with difficult resuscitation or death during use in obstetrical patients.
Patients with hepatic or renal impairment may also require dose adjustments, as these conditions can affect the metabolism and excretion of bupivacaine. Healthcare providers should carefully evaluate each patient’s medical history and current health status before administering bupivacaine and adjust the dose accordingly.
Billing and Coding Considerations
For healthcare providers and facilities, proper billing and coding for bupivacaine administration is essential for reimbursement. The bupivacaine injection cpt code varies depending on the type of procedure and the method of administration. For example, the CPT code for peripheral nerve block using bupivacaine would differ from the code for epidural anesthesia.
When billing for bupivacaine administration, healthcare providers must ensure that they use the appropriate CPT codes and include all necessary documentation to support the medical necessity of the procedure. This includes detailed documentation of the patient’s condition, the procedure performed, the amount and concentration of bupivacaine used, and the patient’s response to the anesthesia.
For facilities that order bupivacaine injection online, understanding the billing and coding requirements is essential for financial viability. Proper coding ensures appropriate reimbursement for the medication and the professional services associated with its administration.
Storage and Handling Guidelines
Proper storage and handling of bupivacaine are essential to maintain its stability and efficacy. Bupivacaine hydrochloride injection should be stored at controlled room temperature, between 20°C to 25°C (68°F to 77°F), with brief excursions permitted between 15°C to 30°C (59°F to 86°F).
The medication should be protected from light and stored in its original container until use. Healthcare providers should inspect the solution for discoloration or particulate matter before administration. Bupivacaine solutions should not be used if they are discolored or contain precipitates.
For facilities that buy bupivacaine injection in USA, implementing proper storage and handling protocols is essential to ensure medication safety and efficacy. This includes regular monitoring of storage temperatures, proper inventory management to prevent expiration, and adherence to aseptic techniques when preparing and administering the medication.
Regulatory and Legal Considerations
As a prescription medication, bupivacaine is subject to regulatory and legal considerations that healthcare providers and facilities must adhere to. In the United States, bupivacaine is classified as a prescription drug, meaning it can only be dispensed with a valid prescription from a licensed healthcare provider.
For medical facilities looking to order bupivacaine injection online, it is essential to ensure that they are working with licensed and legitimate suppliers. The best place to order bupivacaine injection is through authorized medical distributors or pharmacies that comply with all regulatory requirements.
When considering where to buy bupivacaine injection, healthcare facilities should verify the credentials of suppliers and ensure that all necessary documentation is in place. This includes maintaining proper records of purchases, storage conditions, and administration to comply with regulatory requirements and ensure patient safety.
For those who need to order bupivacaine injection in Colorado or any other specific location, it is important to be aware of any state-specific regulations that may apply. Working with knowledgeable legal and compliance professionals can help ensure that all regulatory requirements are met.
Procurement Best Practices
For healthcare facilities, establishing best practices for procuring bupivacaine is essential to ensure quality, safety, and cost-effectiveness. When deciding to bupivacaine injection buy, facilities should consider several factors, including product quality, supplier reliability, pricing, and delivery schedules.
The procurement process should include thorough vetting of potential suppliers, verification of product authenticity, and comparison of pricing from multiple vendors. Facilities should establish clear specifications for the bupivacaine products they require, including concentration, formulation, and packaging requirements.
For those seeking to order bupivacaine injection online, it is crucial to work with reputable suppliers that specialize in medical products. The best place to order bupivacaine injection is one that provides authentic products, competitive pricing, and reliable delivery, while maintaining full compliance with all regulatory requirements. Establishing long-term relationships with trusted suppliers can ensure consistent access to high-quality bupivacaine products.
Frequently Asked Questions (FAQ) About Bupivacaine Injection
1. What are the primary bupivacaine hydrochloride injection uses?
Bupivacaine hydrochloride injection uses are extensive and varied, making it a cornerstone of regional anesthesia. It is primarily indicated for local or regional anesthesia or analgesia for surgical, dental, and oral surgery procedures. It is also widely used for diagnostic and therapeutic procedures, and for obstetrical procedures (excluding the 0.75% concentration). Clinicians use it for infiltration anesthesia, peripheral nerve blocks, sympathetic nerve blocks, epidural blocks, caudal blocks, and retrobulbar blocks. Its long duration of action makes it particularly valuable for post-operative pain management, reducing the need for systemic opioids.
2. What is the difference between bupivacaine 0.25 2.5 mg ml injection and bupivacaine 0.5 injection?
The primary difference between bupivacaine 0.25 2.5 mg ml injection and bupivacaine injection 0.5 is the concentration of the active ingredient, which directly impacts the duration and intensity of the nerve block. The 0.25% (2.5 mg/mL) solution is typically used for procedures where moderate pain relief is sufficient, such as infiltration anesthesia or less intensive peripheral nerve blocks. The bupivacaine injection 0.5 (0.5% or 5 mg/mL) provides a more profound and significantly longer-lasting sensory blockade, making it the preferred choice for major nerve blocks, epidural anesthesia, and surgical procedures requiring extended postoperative pain control. The choice between them depends entirely on the clinical situation, the type of block, and the desired duration of effect.
3. What is the bupivacaine injection cpt code for billing?
The bupivacaine injection cpt code is not a single, universal code but depends entirely on the procedure being performed. The Current Procedural Terminology (CPT) code reflects the medical service, not the specific drug used. For example:
- For a simple peripheral nerve block, codes might range from 64400-64530 depending on the specific nerve.
- For an epidural injection, codes like 62320 (cervical/thoracic) or 62321 (lumbar/sacral) might be used.
- For a trigger point injection, the code is typically 20552 (single or multiple trigger points) or 20553 (multiple trigger points).
The medication itself (bupivacaine) is billed separately using a specific J-code for the drug and the quantity administered. It is crucial for healthcare providers to consult the latest CPT code guidelines and use the specific code that accurately describes the procedure performed.
4. How can I safely order bupivacaine injection online?
To safely order bupivacaine injection online, you must be a licensed medical facility or practitioner. The process requires navigating a regulated supply chain. The safest method is to go through established, authorized medical distributors or licensed pharmacies that require proof of your medical license and a valid prescription or purchase order. When searching online, be extremely cautious of any vendor that does not follow these strict verification procedures. The best place to order bupivacaine injection is always a supplier that is transparent about their credentials, requires proper licensing, and provides full product documentation, including lot numbers and expiration dates.
5. Where is the best place to order bupivacaine injection?
The best place to order bupivacaine injection is from a reputable, licensed medical supplier or pharmaceutical distributor with a proven track record of serving healthcare institutions. These suppliers ensure product authenticity, proper storage and handling (maintaining the cold chain if necessary), and compliance with all regulatory standards. They offer traceability from the manufacturer to your facility, which is critical for patient safety and quality control. For those who need to buy bupivacaine injection in USA, working with large, well-known distributors or specialty pharmacies that specialize in anesthetic agents is the recommended practice.
6. Is it legal to buy bupivacaine injection in Colorado for my clinic?
Yes, it is legal to order bupivacaine injection in Colorado for your clinic, provided you are a licensed medical professional or facility operating within the scope of your practice. You must comply with both federal and state regulations. This means you must have a valid medical license, the medication must be ordered with a legitimate prescription or purchase order for a clinical purpose, and it must be sourced from a licensed pharmaceutical distributor. State-specific regulations in Colorado may also apply, so it is advisable to be familiar with the Colorado State Board of Pharmacy rules regarding the procurement and storage of controlled substances and prescription medications.
7. What are the risks of using bupivacaine and kenalog injection?
Combining bupivacaine and kenalog injection (or other corticosteroids like triamcinolone) is a common practice for therapeutic pain injections, but it carries specific risks. The bupivacaine provides immediate local anesthesia, while the kenalog (triamcinolone acetonide) offers a long-acting anti-inflammatory effect. The risks include those inherent to both drugs: potential for allergic reactions, infection at the injection site, and the specific risks of corticosteroids (e.g., localized tissue atrophy, hyperglycemia, adrenal suppression). There is also a risk of inadvertent intravascular injection, which could lead to systemic toxicity from the bupivacaine. These combinations should only be administered by trained healthcare professionals in a controlled clinical setting.
8. How should I store bupivacaine hydrochloride injection?
Bupivacaine hydrochloride injection should be stored at controlled room temperature, typically between 20°C to 25°C (68°F to 77°F), with brief excursions permitted between 15°C to 30°C (59°F to 86°F). It must be protected from light and kept in its original container. Before administration, the solution should be visually inspected for particulate matter and discoloration. Any solution that is not clear or contains precipitates should be discarded. Proper storage is critical to maintain the medication’s sterility and efficacy, a key consideration for any facility that makes a bupivacaine injection buy.
Conclusion: A Commitment to Excellence in Anesthetic Care
Bupivacaine hydrochloride injection remains an indispensable agent in the modern medical arsenal, offering unparalleled duration and quality of regional anesthesia. Its versatility, from the bupivacaine 0.25 2.5 mg ml injection for minor procedures to the bupivacaine injection 0.5 for major surgical blocks, allows clinicians to tailor pain management strategies to the specific needs of each patient. The development of combination formulations, such as bupivacaine and kenalog injection and bupivacaine and triamcinolone injection, has further expanded its therapeutic utility, particularly in the field of pain management.
However, with this great power comes profound responsibility. The safe and effective use of bupivacaine demands a comprehensive understanding of its pharmacology, meticulous attention to dosage and administration techniques, and the ability to rapidly recognize and manage adverse events. The decision to buy bupivacaine injection is a commitment to upholding the highest standards of patient care, which begins with sourcing the product from legitimate and reputable suppliers.
For healthcare providers and administrators, the procurement process is a critical component of clinical excellence. Knowing where to buy bupivacaine injection is not merely a logistical question but a matter of patient safety. Whether you are looking to order bupivacaine injection in Colorado or need to buy bupivacaine injection in USA for a large hospital system, the principles remain the same: prioritize authenticity, regulatory compliance, and supplier reliability. The best place to order bupivacaine injection is one that guarantees the integrity of its products from manufacturer to patient.
This guide has served as a comprehensive resource, addressing the clinical, administrative, and regulatory facets of using bupivacaine. We have explored its diverse applications, clarified the nuances of different formulations like bupivacaine 0.5 injection, and provided clear guidance on navigating the complexities of procurement. The FAQs have addressed the most common and critical questions, from understanding the bupivacaine injection cpt code to knowing how to safely order bupivacaine injection online.
In conclusion, bupivacaine’s role in medicine is defined by its ability to provide profound, long-lasting pain relief, improving patient outcomes and surgical experiences. By embracing a culture of safety, education, and ethical procurement, healthcare professionals can continue to leverage the full potential of this remarkable medication. Your commitment to excellence, from the moment you order bupivacaine injection to its final administration, is a testament to the highest ideals of patient-centered care.
| Quantity | 10 Vails, 20 Vails, 30 Vails, 60 Vails, 90 Vails, 120 Vails, 360 Vails |
|---|
Be the first to review “Buy Bupivacaine Injection Online” Cancel reply
Related products
KETAMINE
KETAMINE
KETAMINE
KETAMINE
KETAMINE
KETAMINE








Reviews
There are no reviews yet.